capabilities
We’re Backed by Experience
At Resolution Medical our extensive capabilities are backed by practical and clinical experience.
We support each project with a proven process flow, designed to guide you to your next milestone, no matter where you find yourself in your design, development, and manufacturing process.
Design. Develop. Manufacture. Faster
Comprehensive initial “fit” inquiry and open book pricing builds trust
Deep technical, clinical & anatomical expertise in market segments accelerates timelines
Concept to development to manufacturing all under one roof
BioSimulation supports our vertical integration to optimize speed




Seamlessly engage with us at any stage of your product lifecycle.
Seamlessly engage with us at any stage of your product lifecycle.
Concept & Product Development
Fast-track Prototyping:
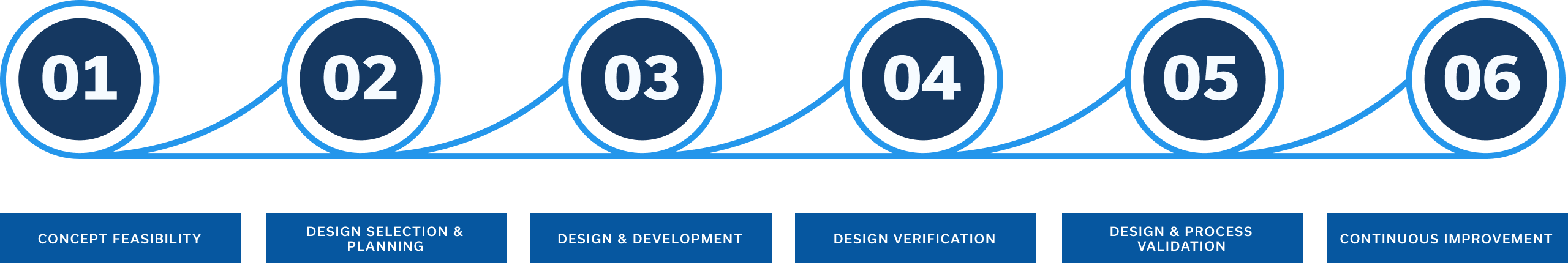
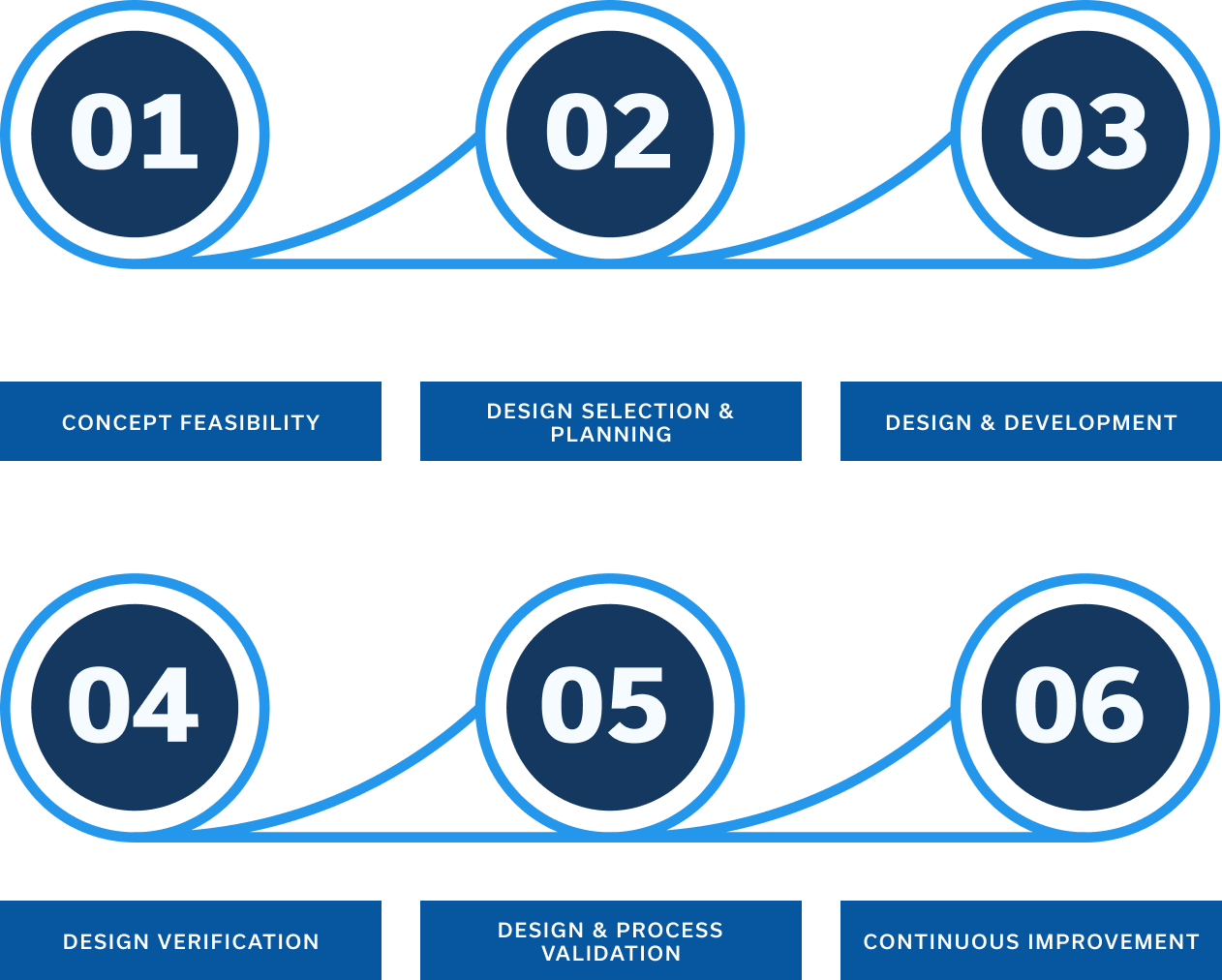
From Concept to Commercialization
Concept Feasibility
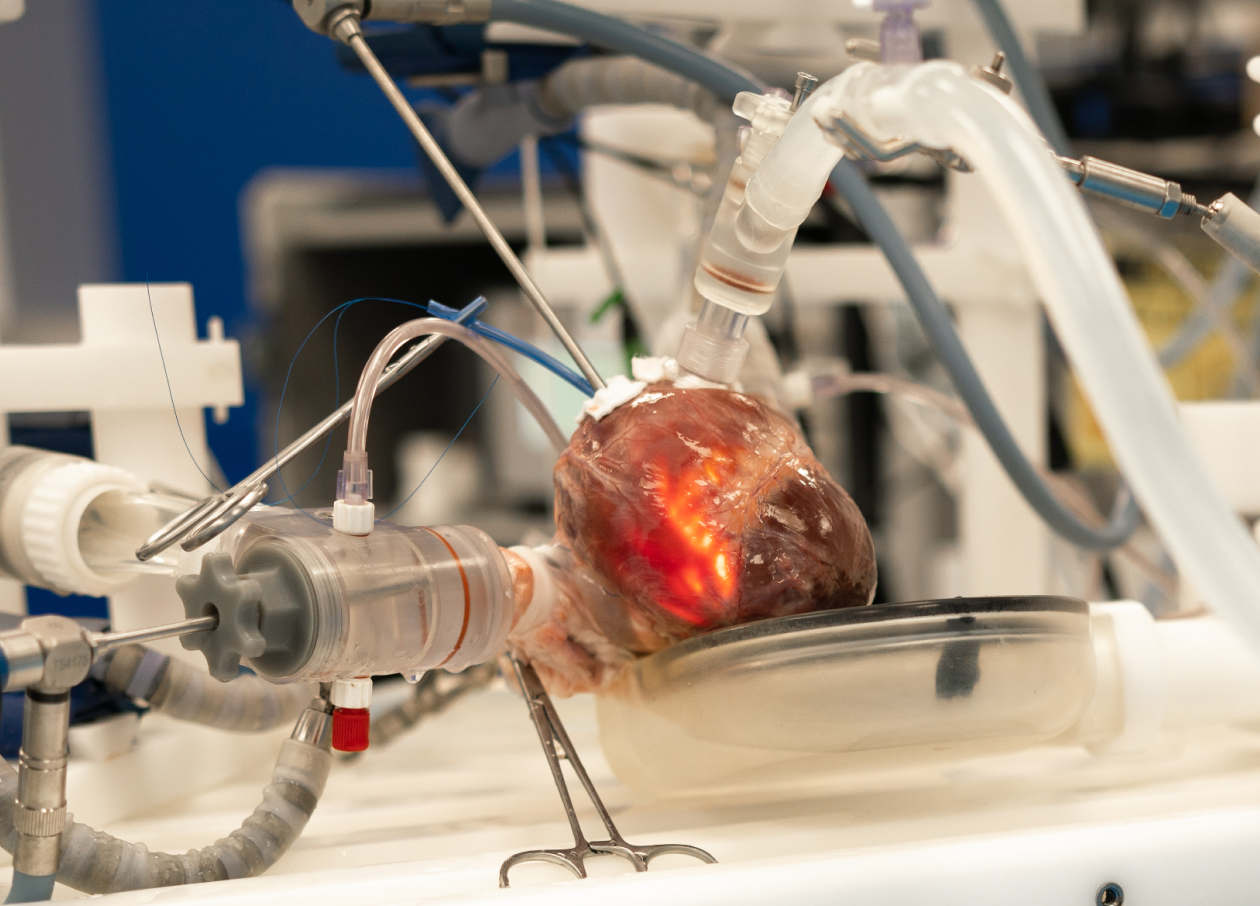
BioSimulation Featuring LifeTec Simulation Technology
Visualize Device Delivery & Deployment in a Pressurized Heart Using BioSimulation
Experience the power of seeing your device in action, iterating, and testing your improvements, all in a single day, expediting development, and saving costs.
We take on even the most difficult challenges and meet them head-on:
Soft Embalming • R&D Acceleration • Clinical Training
SOLUTIONS:
Cardiac BioSimulator, PhysioHeart, BioLiver, Osteochondral Platform,
Heart Valve Assessment Platform, Profused Leg Model, and Vascular Bio Reactor.
Solutions are available for lease at your site, purchase, or in live iteration at our BioSim Lab where we manage the simulator so you can focus on your device.
CAPABILITIES:
Fluoroscopy and ECHO for visualization
Download Our BioSimulation Overview to Learn More

Design Selection & Planning
Design & Development
Design & Process Validation
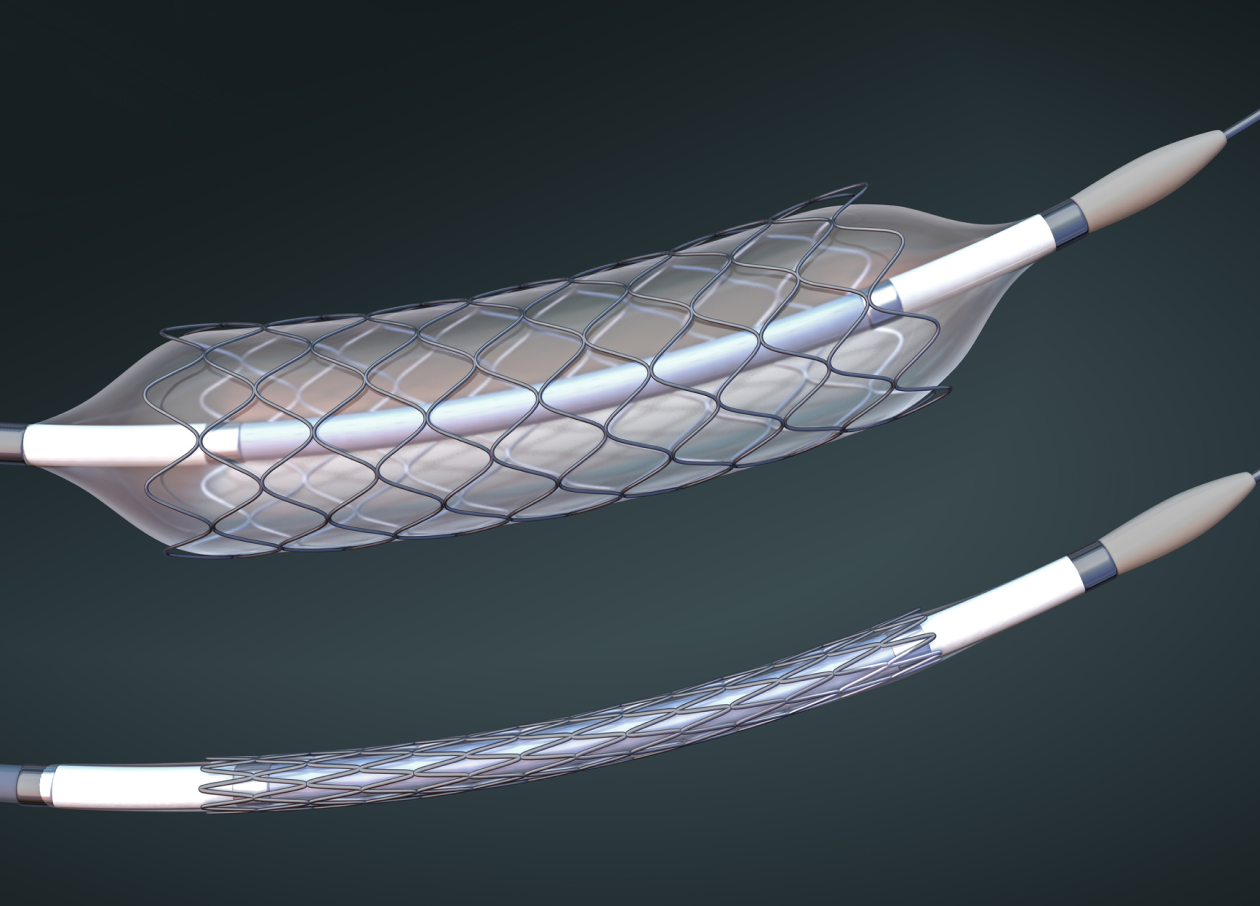
New Product Introduction (NPI) & Process Development
Accelerating Product Launch with Expert NPI
Our dedicated New Product Introduction (NPI) team launches products into manufacturing seamlessly. With experienced process engineers who specialize in design for manufacturing, lean manufacturing, technology transfers, process development, and validation – we’ll guide your transition into manufacturing, whether it’s from design or from an existing manufacturing line.
Navigating the highly regulated landscape of medical device manufacturing can be challenging, but we’re well-versed in the intricacies of regulatory compliance. Our experts anticipate obstacles and guide you toward the most efficient path to success, ensuring compliance and scale.
Download Our Overview to Learn More

Design Selection & Planning
Design & Development

Manufacturing of Sub-Assemblies & Finished Devices
Comprehensive Manufacturing Capabilities for Complex Medical Devices
With a dedicated 36,000 sqft. facility, FDA registration, and decades of experience, we are equipped for low to high volume manufacturing. Our high-touch collaboration process is built on transparency and direct communication. We excel in both cleanroom and non-cleanroom assembly.
Our ISO Class 7 Cleanrooms enable precise finished goods assembly, sterile barrier packaging, product labeling, inspection, and cleaning. We also have an electrostatic discharge area (UL Certified) and a distribution center for efficient logistics.
Download Our Manufacturing Overview to Learn More
- Complex Device Assembly
- Micro Soldering
- Active Implantable Assembly
- Neuro Lead Coiling
- Electro-mechanical Assembly & Testing.
- Multi-balloon Catheter Assembly & Leak Testing
- Sterile Barrier Assembly
- Managing Sterilization
- Compliance & Regulatory Requirements
- Laser Cutting & Post-processing
- Welding
- Metals Processing (Nitinol, Titanium, Stainless, Platinum)
- Plastics Processing (Peek, Nylons, etc.).
- Sub-assemblies, testing, analysis, and hermetic welds in our production Glovebox.
- Additive manufacturing
- Molding services

Concept Feasibility
Design Verification

Specialized Experience
Serving a Wide Range of Markets